barium carbonate precipitated powder |
|
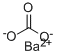 |
|
Item : baco3 |
|
Details :Identification of 513-77-9
Name:Barium carbonate EINECS: 208-167-3 Molecular Formula:BaCO3 CAS Registry Number: 513-77-9 Synonyms: barium carbonate, natural; barium carbonate, other than natural; Barium Carbonate (Powder); Barium Carbonate (Low Sulphur); BariumcarbonateCaSrmeshwhitepowder; Bariumcarbonatewhitepowder; Barium carbonate,( 99.9%-Ba; Barium carbonate 99+ %; Barium Carbonate (free flowing); Barium carbonate,high purity InChI: InChI=1/CH2O3.Ba/c2-1(3)4;/h(H2,2,3,4);/q;+2/p-2 Molecular Structure: This structure is also available as a 2d Mol fileChemical Properties Appearance: White powder Molecular Weight: 197.34 Density: 4.43 Boiling Point: 1450℃ Melting Point: 811℃ Flash Point: 169.8 °C Storage Temperature: Store at RT. Solubility: 0.002 g/100 mL (20 °C) Stability: Stable. Incompatible with strong acids.Safety Data of 513-77-9 Risk Codes: R22 Safety Statements: S24/25 Hazard Symbols: Xn:Harmful HazardClass:6.1(b) ----------------------------------------------------------------------------------------------------------------------------------------- UsesBarium carbonate is widely used in the ceramics industry as an ingredient in glazes. It acts as a flux, a matting and crystallizing agent and combines with certain colouring oxides to produce unique colours not easily attainable by other means. Its use is somewhat controversial since some claim that it can leach from glazes into food and drink. To provide a safe means of use, BaO is often used in fritted form. In the brick, tile, earthenware and pottery industries barium carbonate is added to clays to precipitate soluble salts (calcium sulfate and magnesium sulfate) that cause efflorescence. |
|
| [Previous15630-89-4] | |
|

